Ensuring that a manufacturer is FDA-certified is crucial for businesses in the Philippines. But what does it mean to be an FDA-certified manufacturer? Simply put, it means that the manufacturer follows strict guidelines set by the Philippine FDA to ensure product safety and quality. This certification is essential for industries such as food, drugs, cosmetics, and medical devices. Companies that are not certified risk facing legal penalties and losing consumer trust. OSPA Korean Beauty Corp, a leading FDA-certified manufacturer of raw materials and empty gelatin capsules in the Philippines, understands the importance of compliance and quality.
Guide to Checking FDA Certified Manufacturer
When looking for a reliable supplier, businesses often wonder how to verify if a manufacturer is truly FDA-certified. This involves checking for a License to Operate (LTO) and a Certificate of Product Registration (CPR), which are issued by the Philippine FDA. These documents are proof that the manufacturer meets the required quality parameters and follows Good Manufacturing Practices (GMP). But how do you ensure that these certifications are genuine? This is where understanding the process and asking the right questions come into play.
What Does It Mean to Be an FDA-Certified Manufacturer in the Philippines?
Being an FDA-certified manufacturer in the Philippines means adhering to strict regulations that ensure products are safe for consumption or use. The Philippine FDA plays a vital role in overseeing these industries to protect public health. Manufacturers must comply with GMP, which involves maintaining clean facilities, using quality raw materials, and following precise production processes. This not only ensures product quality but also helps prevent contamination and defects.
Key Aspects of GMP Compliance
- Clean Facilities: Manufacturers must maintain clean and hygienic facilities to prevent contamination.
- Quality Raw Materials: Only high-quality raw materials are used to ensure product safety and efficacy.
- Precise Production Processes: Strict protocols are followed during production to maintain consistency and quality.
For instance, when it comes to raw materials and gelatin capsules, manufacturers must ensure that they meet specific standards. This includes sourcing high-quality ingredients and maintaining precise manufacturing conditions. OSPA Korean Beauty Corp, with its extensive range of raw materials sourced from countries like Korea and Japan, demonstrates a commitment to quality. Their customizable gelatin capsules cater to various needs, from pharmaceutical to veggie capsules, showcasing flexibility and innovation.
Steps to Verify an FDA-Certified Manufacturer in the Philippines
Verifying if a manufacturer is FDA-certified involves several steps. First, check if they have a valid LTO and CPR. This can be done by contacting the Philippine FDA directly or checking their official databases. It’s also important to inspect the manufacturing facility to ensure it meets GMP standards. This includes checking for cleanliness, proper equipment maintenance, and adherence to production protocols.
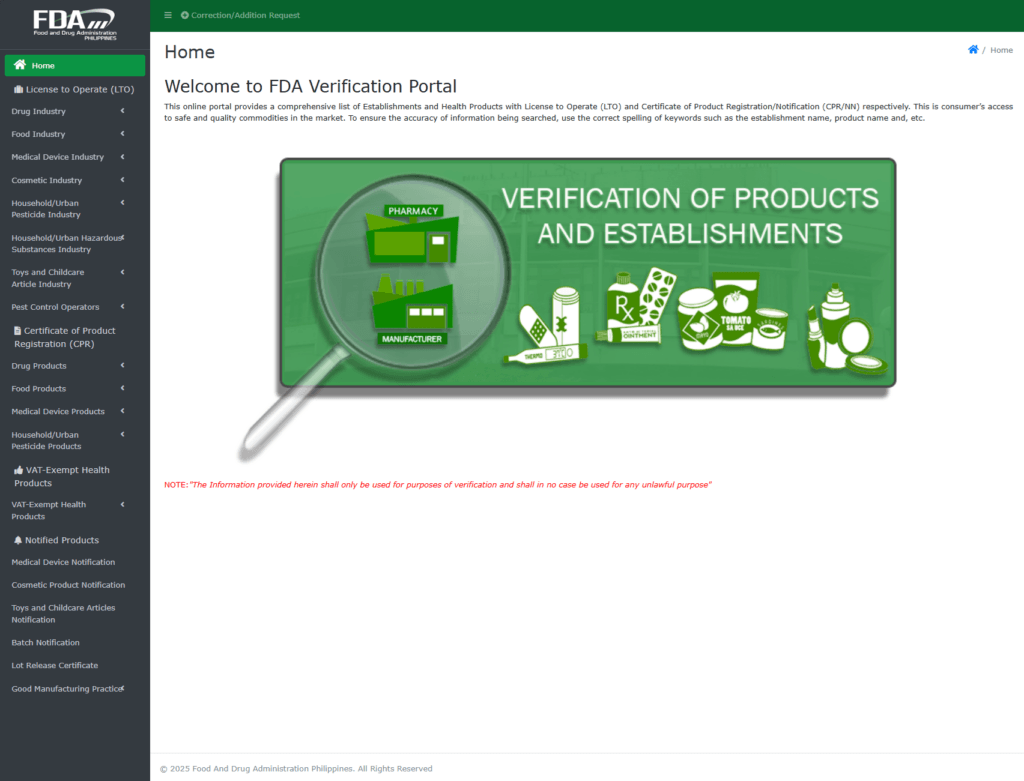
How to Verify Certifications
- Check Official FDA Databases: Verify the manufacturer’s certifications through official channels.
- Inspect Manufacturing Facilities: Ensure facilities meet GMP standards by conducting site visits.
- Request Documentation: Ask for copies of the LTO and CPR to confirm authenticity.
When verifying GMP compliance, look for a valid GMP certificate. This certificate indicates that the manufacturer has passed rigorous inspections and maintains high-quality manufacturing processes. OSPA Korean Beauty Corp is transparent about their certifications and welcomes inspections to ensure compliance.
Why Choose OSPA Korean Beauty Corp as Your Trusted Manufacturer?
OSPA Korean Beauty Corp stands out as a premier FDA-certified manufacturer in the Philippines. We specialize in providing over 10,000 high-quality raw materials and customizable empty gelatin capsules. Our commitment to quality is evident in their ISO-certified silica gel products, which are used in medicine packaging. What sets us apart is their ability to deliver products quickly, often within 30 days, while maintaining stringent quality standards.
When choosing a manufacturer, businesses should ask questions about their quality control processes, certification status, and product customization options. Here are some key questions to ask your manufacturer:
- What certifications do you hold?
- How do you ensure product quality?
- Can you provide references from previous clients?
By asking these questions, businesses can ensure they are partnering with a reliable and compliant supplier.
Common Challenges in Verifying FDA Certification
Verifying FDA certification can be challenging. One common issue is counterfeit certifications. Some manufacturers might claim to be certified when they are not. To avoid this, it’s crucial to verify certifications directly with the Philippine FDA. Another challenge is incomplete documentation. Manufacturers might not provide all necessary documents, which can raise suspicions about their compliance.
Overcoming Verification Challenges
- Verify Certifications Directly: Contact the Philippine FDA to confirm the authenticity of certifications.
- Request Complete Documentation: Ensure all necessary documents are provided to verify compliance.
- Conduct Site Visits: Inspect facilities personally to ensure they meet GMP standards.
To overcome these challenges, businesses should be proactive. They should request all relevant certifications and inspect manufacturing facilities personally. OSPA Korean Beauty Corp encourages transparency and welcomes clients to inspect their facilities, ensuring trust and compliance.

The Importance of Partnering with an FDA-Certified Manufacturer
Partnering with an FDA-certified manufacturer is vital for any business. It ensures that products meet quality parameters and are safe for consumers. Non-compliance can lead to severe legal penalties and damage to a company’s reputation. By working with certified manufacturers like OSPA Korean Beauty Corp, businesses can ensure they are producing high-quality products that meet regulatory standards.
Moreover, certified manufacturers provide peace of mind. They have systems in place to prevent contamination and ensure consistent quality. This not only protects consumers but also helps businesses maintain a positive reputation in the market. In a competitive industry, partnering with an FDA-certified manufacturer can be a strategic advantage.
Conclusion
In conclusion, verifying if a manufacturer is an FDA-certified manufacturer in the Philippines involves checking for key certifications like an LTO and CPR, ensuring GMP compliance, and asking the right questions. OSPA Korean Beauty Corp exemplifies what it means to be a reliable and compliant supplier, offering high-quality raw materials and customizable gelatin capsules. By partnering with such manufacturers, businesses can ensure they are producing safe and compliant products, which is essential for success in today’s competitive market.














